What metal does not dissolve in acid?
Copper Insoluble in typical strong acids because the overall cellular potential for copper metal oxidation to Cu2+ ions along with H+ ion reduction to H2 is negative.
Which metal is insoluble in acids?
A group of elements known as precious metals or precious metals dissolve only the hard way. In particular, platinum, iridium, gold and osmium stand up to the attack of strong hydrochloric and nitric acids.
Which metal does not react easily with acids?
Most metals react with dilute acids to form their respective salts along with hydrogen gas. But Copper ( ), silver ( ) and mercury ( ) do not react with dilute acids.
Which metal is insoluble in HCL?
The metals copper and mercury do not react with dilute hydrochloric acid because they are behind hydrogen in the active series, i.e. they cannot displace hydrogen from hydrochloric acid.
See also How old is Carl when he dies?Which substance is insoluble in acid?
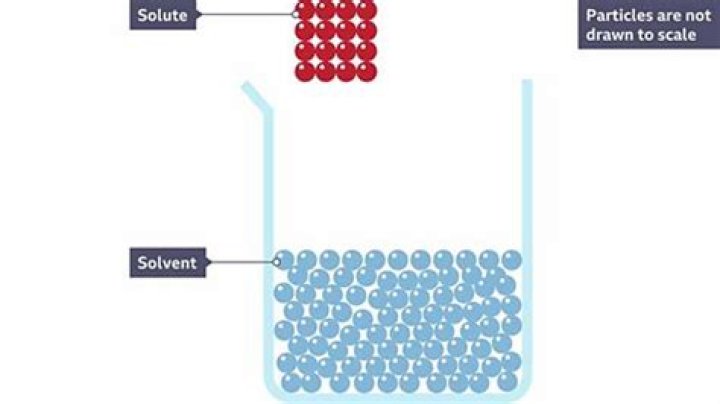
Strong acids will attack many metals, turning them into soluble ions and releasing bubbles of hydrogen gas in the process. Acids will also dissolve carbonates such as limestone, and some other minerals and inorganic compounds.
Metallic sodium and water
What cannot react with acids?
Acids do not react with metals except Zn, Al and Sn.
Which acid can dissolve steel?
Hydrochloric acid is so strong it can corrode metals that you can see it firsthand in the school chemistry lab.
Which metal is most resistant to hydrochloric acid?
Zirconium and its alloys have strong corrosion resistance to most hydrohalic acids, especially hydrochloric acid, bromhydric acid, hydroiodic acid and hypochlorous acid.[18]. Corrosion rate less than 0.13mm/a at any atmospheric hydrochloric acid concentration, boiling point or higher temperature[19].
Can HCl dissolve aluminum?
At room temperature, aluminum reacts with dilute hydrochloric acid. Colorless aluminum chloride and hydrogen gas are produced when the metal dissolves in hydrochloric acid.
Will HCl dissolve stainless steel?
Hydrochloric acid is an exception to the general acid resistance of stainless steel and should be avoided.
What are two metals that do not react with acids?
We can understand from the above discussion that the gold metal does not react with acids. Therefore, the correct answer is C Yellow.
Do all metals react with acids?
Many, but not all, metals react with acids. Hydrogen gas is formed when metals react with acids to form salts. Many, but not all, metals react with acids. Hydrogen gas is formed when metals react with acids to form salts.
See also How are prisoners on TikTok?Does aluminum react with acids?
Answer: Aluminum reacts with both acids and alkalis. When it reacts with an acid, it produces aluminum chloride and hydrogen gas.
Which metal is resistant to all acids?
High Silicon—High silicon irons such as Duriron and Corrosiron exhibit excellent resistance to all concentrations of sulfuric acid up to and including boiling point.
Which metal reacts violently with acids?
Metals such as potassium, sodium and lithium react strongly with acids. This is because a lot of heat energy is released during the reaction, causing the hydrogen to explode. Less reactive metals such as magnesium, zinc and iron react less explosively.
Does copper react with acids?
in the case of copper Copper is lower than hydrogen in the chain reaction. This means that copper is less reactive than hydrogen and therefore cannot push it out of an acidic solution. so this is the reason copper doesn’t react with hydrochloric acid.
Does titanium react with acids?
Reaction of titanium with acids
Titanium metal does not react with inorganic acids at room temperature but reacts with hot hydrochloric acid to form titanium(III) complexes.
Does gold react with acids?
Gold is unaffected by air, water, alkali and all acids except aqua regia (a mixture of hydrochloric acid and nitric acid) which can dissolve gold. In fact, gold’s resistance to acid is one of the reasons why our acid test is so accurate.
Which acid eats aluminum?
Sulfuric acid dissolves aluminum metal according to the reaction: 2 Al(s) + 3 H2SO4(aq)¡Al2(SO4)3(aq) + 3 H2(g) Suppose you want to dissolve a mass of aluminum 15.2 g .
See also How do you know if you have a good ground?What does acid do to stainless steel?
acid. Strong acids destabilize the passivation layer. When stainless steel is exposed to hydrochloric and sulfuric acids, general surface corrosion may occur. Hydrochloric acid is a very strong acid and is used in the manufacture of plastics.
What is most resistant to acids?
PTFE (or Teflon™) – PTFE, also known as Teflon™, is one of the best acid resistant materials available and is commonly used in chemical and pharmaceutical laboratory applications.
What is Acid Resistant Stainless Steel?
Stainless steel contains mainly iron with a minimum of 10.5% chromium (according to: EN 10020). Steel with a chromium content of at least 18% is called acid resistant steel.
Does rust dissolve in acid?
Strong acids will dissolve rust, but they will also dissolve paint, finishes, and sometimes metal. Hydrochloric acid (also known as muriatic acid in diluted form), as well as phosphoric and sulfuric acids can be used in strong acid rust removal formulations.
Can acids melt diamonds?
No, acids cannot dissolve diamond, for the simple reason that the diamond’s carbon atoms are too tightly bound together for Hydrogen ions to dissolve the substance.
Can acids eat through metals?
Strong acids usually dissolve metals, but some metals are stable enough to resist the effects of acids. Aqua regia means “royal water” because this mixture of hydrochloric acid and nitric acid can dissolve precious metals, such as gold and platinum. Acids themselves cannot dissolve these metals.
Let the article source What metal does not dissolve in acid? of website
Categories: Frequently Asked Questions